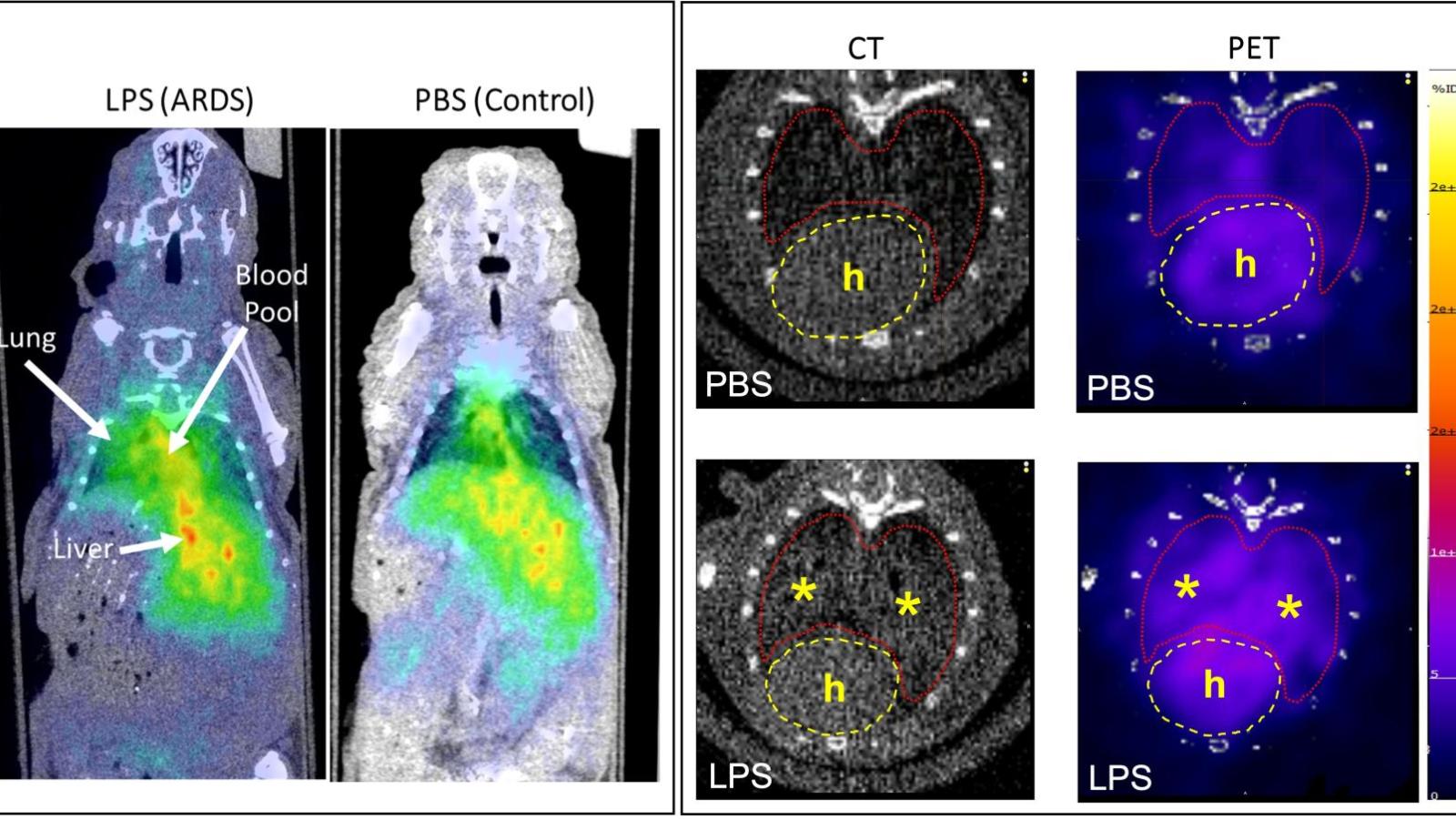
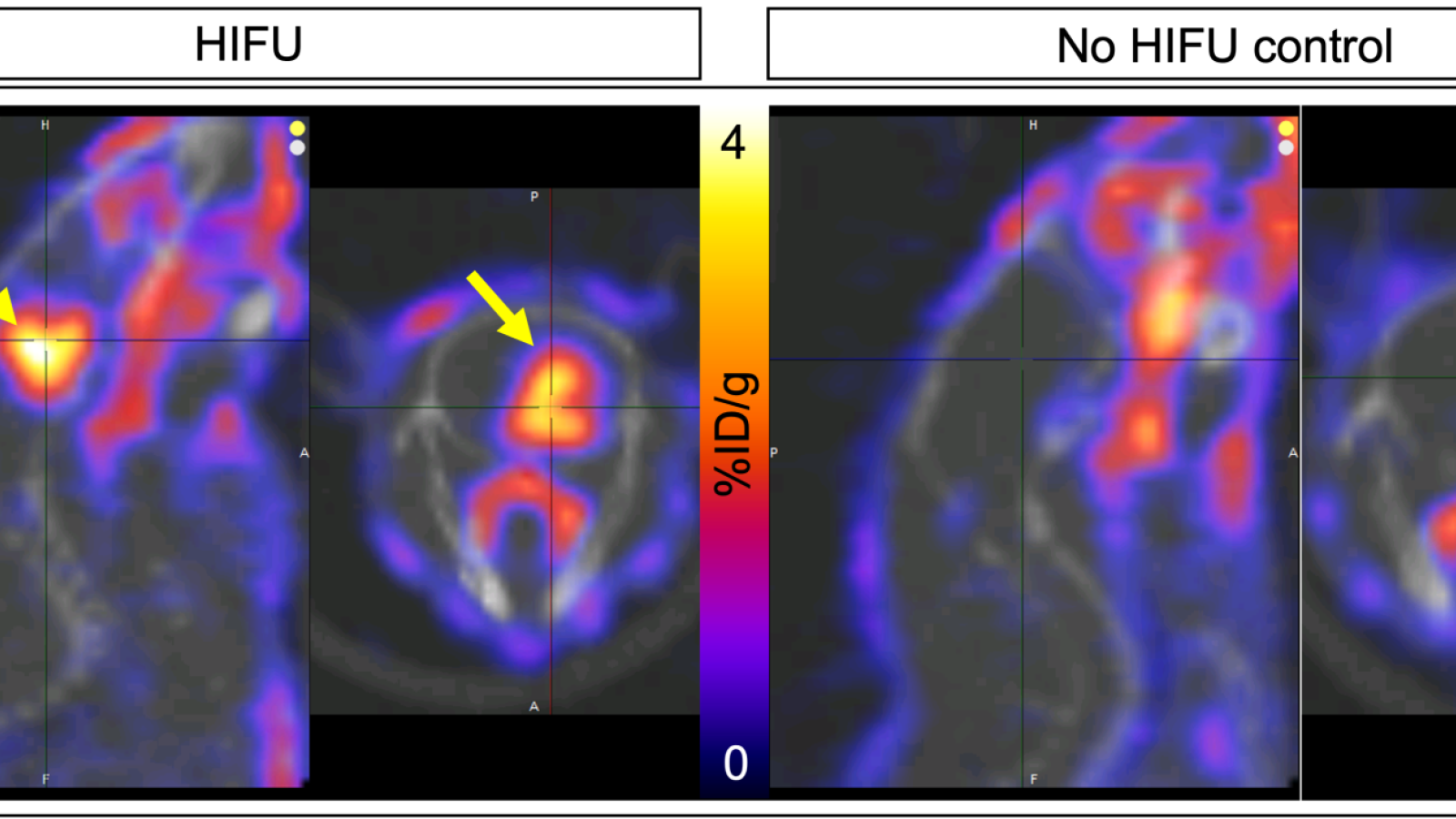
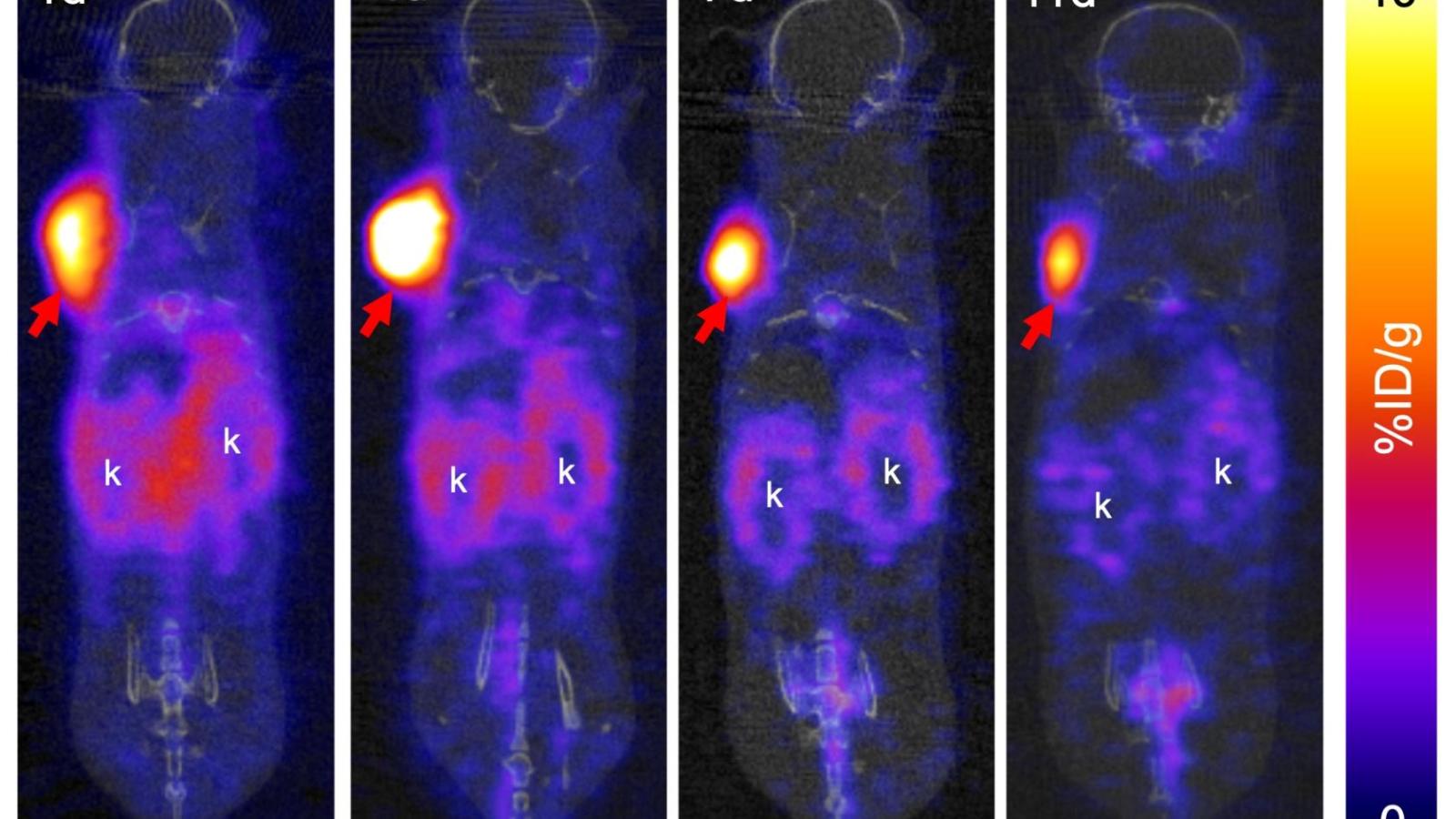
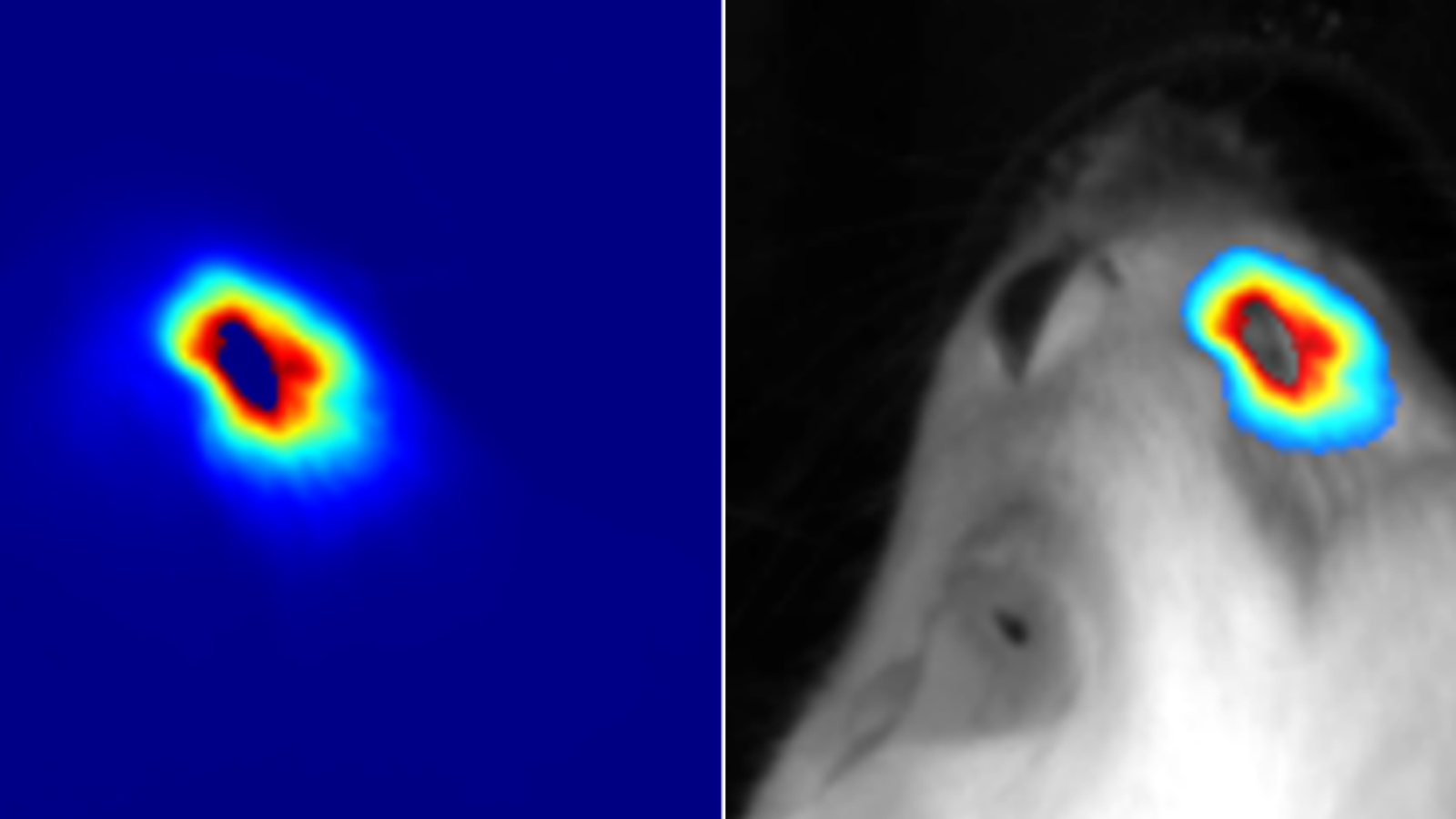
Radiopharmaceutical Translation Program
The Columbia University Radiopharmaceutical Translation Program is a close collaboration between the Department of Radiology, Herbert Irving Comprehensive Cancer Center (HICCC) Experimental Therapeutics/Phase 1 Program, and scientific expertise at Columbia University. Our program develops radioisotopes and translates radioisotopes developed at Columbia and pharmaceutical companies, to advance the next generation of radiopharmaceutical agents.
We have deep technical and regulatory expertise as well as state-of-the-art facilities required to translate novel diagnostic and therapeutic agents from bench-to-bedside including:
- A full radiochemistry development program
- PET cGMP radiotracer manufacturing, holding aNDAs, and INDs for clinical and research use
- Clinical imaging assets including PET/CT, SPECT/CT and MRI scanners
- Dedicated clinical trial personnel with expertise in early phase radiopharmaceutical development
- Interdigitation with the HICCC Experimental Therapeutics/Phase 1 Program to harness the operational and regulatory expertise of the NCI-funded HICCC Clinical Protocol and Data Management (CPDM) resources
- Close partnerships with the HICCC Disease Related Groups (DRGs)
- Preclinical laboratories and imaging facilities specialized for use with radioactivity, including MicroPET, microCT, Optical/Bioluminescent scanners; a dedicated satellite small animal facility to house radioactive preclinical experiments; and a full Cell and Molecular Biology laboratory with a dedicated flow cytometer, cryostat, advanced liquid handling systems, and other equipment.
- Expertise in radiopharmaceutical and immune system interactions
Contact
For more information and collaboration inquiries, please contact:
Stella Kang, MD, MS
PETresearch@cumc.columbia.edu